 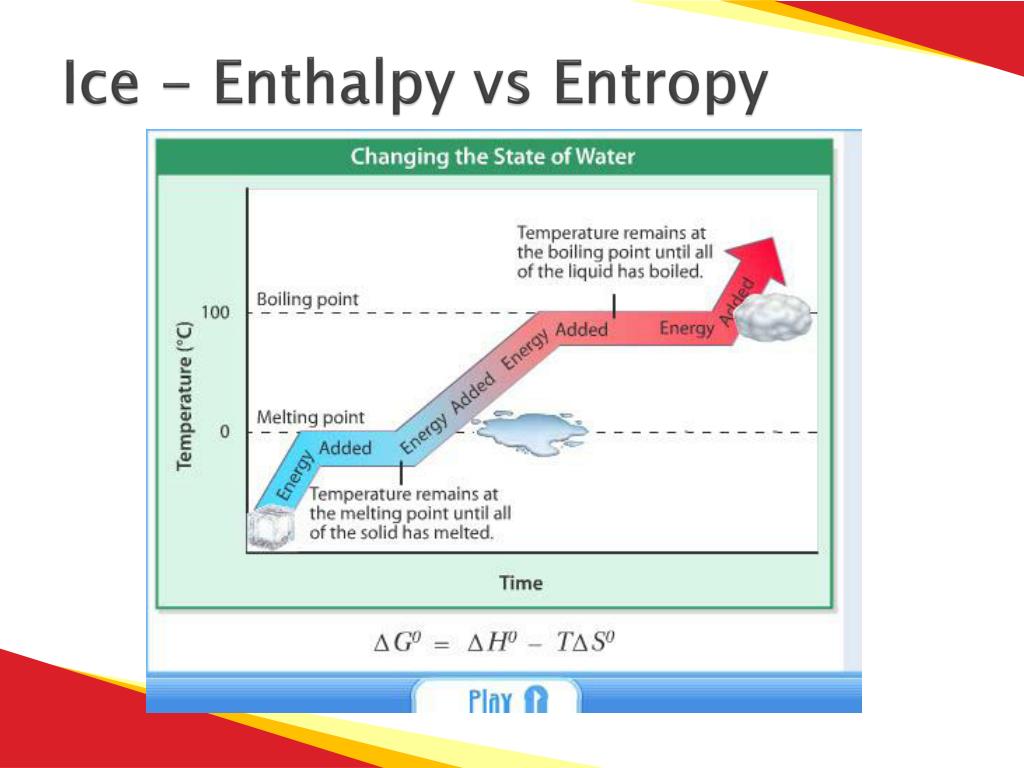
Enthalpy is denoted as ‘ΔH.’ It tells about the difference between the enthalpy of the products and the enthalpy of the reactants. Enthalpy is the measure of total energy in the system, although it always denotes the change in the energy system at constant pressure as it as the total enthalpy of the system can’t be measured. Enthalpy - Is There a Difference?Įnthalpy measures the heat change or internal energy change of a system during the chemical reaction under constant pressure. The temperature in the entropy equation is measured on the absolute or Kelvin temperature scale. The SI unit for Entropy (S) is Joules per Kelvin (J/K). In an equation, it is written as ‘ΔS’ as it represents the disorder in the entropy during the chemical process. 'S’ denotes entropy, and it is always written as a capital letter. The measure of that change of energy or disorder during the chemical process is entropy. It can be mechanical, chemical, thermal, nuclear or any other energy. As we know that energy in a body tells about its capability to do work, the energy in a body can be of any type. Actually, it is the measure of random activity, which is usually the amount of disorder in the system. What is Entropy?Entropy is the measurement of the disorder or the randomness in the system during the chemical process. On the other hand, entropy is the measure of random activity, which is usually the amount of disorder in the system. Actually, enthalpy is the measure of total energy in the system, although it always denotes the change in the energy system at constant pressure as it as the total enthalpy of the system can’t be measured. Entropy is the measurement of the disorder or the randomness in the system during the chemical process, whereas enthalpy measures the heat change or internal energy change of a system during the chemical reaction under constant pressure. Entropy and Enthalpy are the famous terms related to thermodynamics. This also deals with the relations of heat with other forms of energy such as electrical, mechanical or chemical energy. Thermodynamics is the branch of physics that deals with the study of heat and other relating phenomena. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
